Resource
- Why Special Handling Matters (Pre-Analytical Quality)
- The 4 Core Reasons Specimens Are Sensitive (The “Why” Behind Special Handling)
- Tests That Require Special Handling: The Main Categories
- 1) Temperature-Sensitive Tests
- A. Must be kept cold (on ice or chilled rapidly)
- B. Must be refrigerated (2–8°C)
- C. Must be frozen (−20°C / −70°C)
- 2) Light-Sensitive Tests (Protect From Light)
- 3) Time-Critical Tests (Rapid Transport / Immediate Processing)
- 4) Tests Requiring Specific Tubes, Additives, or Fill Ratios
- 5) Anaerobic Handling (Avoid Air Exposure)
- 6) Microbiology & Molecular Tests: Sterility, Media, and Contamination Control
- 7) Histopathology & Cytology: Fixatives and Identity Control
- 8) Genetics, Toxicology, and Forensics: Chain-of-Custody
- 9) High-Risk Specimens: Biosafety and Exposure Prevention
- A Practical “Special Handling” List by Specimen Type (Easy to Train)
- A) Blood (Whole Blood / Serum / Plasma)
- B) Urine
- C) Swabs / Respiratory Specimens
- D) CSF / Body Fluids (CSF, pleural fluid, ascites, etc.)
- Transport & Packaging: The Most Common Failure Point
- Special Handling Also Depends on Lab Setup (Workflow + Hardware)
- 1) Specimen Receiving Area (Sample Intake)
- 2) Cold Chain Storage Planning
- 3) Safety & Ventilation
- A Simple “Special Handling” Checklist (Print This)
- FAQ
- 1) What’s the most common reason a lab sample is rejected?
- 2) Which tests are most sensitive to transport delays?
- 3) Why do some samples need protection from light?
- 4) Do all PCR/molecular tests require freezing?
- 5) How can a lab reduce special-handling errors quickly?
- 6) What tests require “keep warm” handling?
- 7) What is the difference between refrigerated transport and “on ice” transport?
- 8) How do I know whether a test needs light protection?
- 9) Can poor handling cause false positives?
- 10) What should a small diagnostic lab prioritize first?
Which Laboratory Tests Require Special Handling? A Practical Guide to Temperature, Timing, Light, and Safety-Critical Specimens
Many “abnormal” lab results are caused by poor specimen handling—not the patient’s condition. This in-depth guide explains which laboratory tests require special handling, why specimens are sensitive, and how to control temperature, time, light, packaging, and biosafety. You’ll also learn how a specimen-friendly lab setup (receiving, storage, wet area, ventilation) reduces errors and improves quality.
Why Special Handling Matters (Pre-Analytical Quality)
Before any sample reaches an analyzer, it can be altered by heat, cold, light, delays, agitation, wrong collection tubes, contamination, or air exposure. This is the pre-analytical phase—and it’s where a large share of preventable lab errors occur.
“Special handling” means a specimen has extra requirements such as:
-
Temperature control (ice, refrigerated, frozen, or controlled warm)
-
Time limits (STAT delivery, rapid processing, immediate separation)
-
Protection from light
-
Specific collection tubes/additives and fill ratios
-
Air exclusion (anaerobic handling)
-
Sterile technique
-
Biosafety precautions
-
Chain-of-custody documentation
Understanding these requirements helps you prevent inaccurate results, reduce specimen rejection, and protect staff.
The 4 Core Reasons Specimens Are Sensitive (The “Why” Behind Special Handling)
If you understand these four mechanisms, special-handling rules become easier to follow and enforce:
-
Ongoing cell metabolism
Blood cells remain active after collection. They can consume glucose, generate lactate, and shift pH/electrolytes if separation is delayed. -
Photodegradation and oxidation
Some analytes break down under light or oxygen (e.g., bilirubin, porphyrins, certain vitamins and drugs). -
Protein/enzyme activity changes with temperature
Warmer temperatures often accelerate degradation or reactions; freezing can preserve unstable compounds for longer transport. -
Contamination and inhibition
Microbiology and molecular tests are vulnerable to contamination (false positives) or inhibitors (false negatives), especially when collection and processing areas are not well controlled.
Tests That Require Special Handling: The Main Categories
1) Temperature-Sensitive Tests

A. Must be kept cold (on ice or chilled rapidly)
These analytes change quickly at room temperature.
Common examples
-
Blood gases (ABG/VBG) (also time/air sensitive)
-
Lactate
-
Ammonia
-
Pyruvate (often very strict)
-
Some peptide hormones (method-dependent)
Best practices
-
Use proper transport materials (ice/chilled packs as required).
-
Minimize delay; deliver immediately.
-
Avoid vigorous shaking; invert gently as required.
B. Must be refrigerated (2–8°C)
Used for short-term stability and delayed processing.
Common examples
-
Many serology specimens (short-term)
-
Certain chemistry specimens when delays are expected (protocol-dependent)
-
Some therapeutic drug monitoring samples (varies)
Best practices
-
Store sealed, upright.
-
Separate serum/plasma from cells within the recommended timeframe when required.
C. Must be frozen (−20°C / −70°C)
For unstable analytes or long transport.
Common examples
-
Viral load PCR (often frozen if delayed)
-
Specialty cytokines / endocrine tests (assay-dependent)
-
Confirmatory toxicology samples (protocol-dependent)
Best practices
-
Freeze promptly if not analyzed soon.
-
Avoid repeated freeze–thaw cycles (aliquot if needed).
-
Use leak-proof vials and secondary containment.
2) Light-Sensitive Tests (Protect From Light)
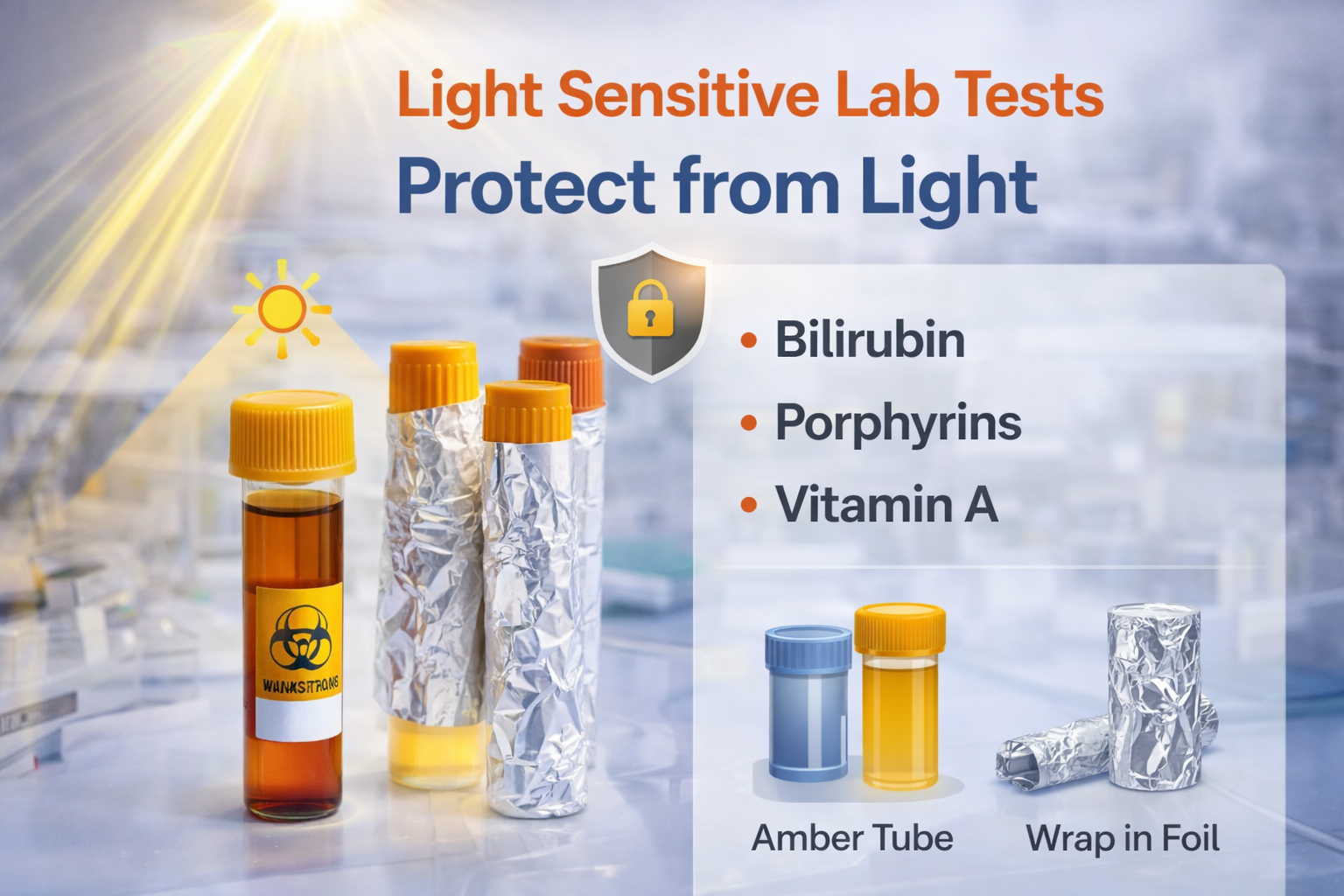
Common examples
-
Bilirubin (especially neonatal bilirubin)
-
Porphyrins
-
Vitamin A and other fat-soluble vitamins (method-dependent)
-
Some photolabile drugs
Best practices
-
Use amber containers or wrap tubes in foil.
-
Keep out of direct sunlight and strong lighting.
-
Transport quickly.
3) Time-Critical Tests (Rapid Transport / Immediate Processing)
Even if temperature is correct, some tests require fast processing.
Common examples
-
Glucose (if not using glycolysis inhibitor and processing is delayed)
-
Potassium (affected by hemolysis and delays)
-
Coagulation tests (PT/INR, aPTT) often have processing time windows
-
Platelet function tests (highly time sensitive)
-
CSF analysis (cell counts & microbiology need speed)
Best practices
-
Use STAT transport when required.
-
Follow tube fill ratios (especially for coagulation).
-
Centrifuge/separate specimens when required and within specified windows.
4) Tests Requiring Specific Tubes, Additives, or Fill Ratios
Wrong collection tubes are a leading cause of specimen rejection and inaccurate results.
Common examples
-
Coagulation: citrate tubes (must be filled to the line)
-
Trace elements/heavy metals (lead, zinc, copper): trace-element–free tubes
-
Drug levels: certain assays require specific tube types (gel separators may interfere in some methods)
-
Blood cultures: sterile bottles with correct volumes
Best practices
-
Train staff on tube selection and order of draw.
-
Enforce fill-volume standards (especially citrate).
-
Standardize supplies where possible to reduce variability.
5) Anaerobic Handling (Avoid Air Exposure)
Common examples
-
Blood gas specimens
-
Anaerobic cultures (certain aspirates/wounds)
Best practices
-
Expel air bubbles from blood gas syringes per protocol.
-
Use anaerobic transport media for culture samples.
-
Seal tightly and transport immediately.
6) Microbiology & Molecular Tests: Sterility, Media, and Contamination Control
In infection testing, incorrect media or contamination can create false results.
Common examples
-
Blood cultures: skin antisepsis + correct volume is critical
-
CSF culture: immediate transport (don’t refrigerate unless instructed)
-
NAAT swabs (e.g., CT/NG): correct swab type + transport medium
-
Stool culture / O&P: correct preservatives and timing
-
Respiratory viral PCR: correct swab + cold chain when delayed
Best practices
-
Use sterile technique and correct containers.
-
Follow transport media requirements exactly.
-
Separate “clean” prep areas from high-copy PCR areas.
-

7) Histopathology & Cytology: Fixatives and Identity Control
Common examples
-
Biopsy specimens: formalin fixation (except when fresh tissue is needed)
-
Frozen section: fresh tissue, immediate delivery
-
Cytology samples: correct preservative vials
Best practices
-
Confirm fixative type and volume (tissue-to-fixative ratio matters).
-
Label at bedside with two identifiers.
-
Use leak-proof containers and follow chemical safety rules for fixatives.
8) Genetics, Toxicology, and Forensics: Chain-of-Custody
Common examples
-
Forensic toxicology
-
Workplace drug testing
-
Identity/paternity genetic testing
-
Certain clinical trials specimens
Best practices
-
Tamper-evident seals and documented handoffs.
-
Controlled access storage.
-
Precise timestamping and witnessed collection when required.
9) High-Risk Specimens: Biosafety and Exposure Prevention
Common examples
-
Suspected TB or high-consequence pathogens
-
Unknown-risk large-volume body fluids
Best practices
-
Follow your biosafety level procedures.
-
Use triple packaging for transport: primary leak-proof container, secondary sealed container with absorbent, rigid outer packaging.
-
Clear hazard labeling and spill-response readiness.
-

A Practical “Special Handling” List by Specimen Type (Easy to Train)
Many labs find specimen-type instructions easier to implement than test-by-test lists.
A) Blood (Whole Blood / Serum / Plasma)
Common special-handling scenarios
-
Blood gas: time sensitive + minimize air exposure
-
Coagulation (PT/INR, aPTT): strict fill ratio and processing windows
-
Therapeutic drug monitoring: collection timing (peak/trough) + storage requirements
-
Lactate/ammonia/some hormones: temperature + delay sensitive
Key handling steps
-
Define the time window from collection → lab receipt → centrifuge → separation → storage.
-
Enforce correct tube type and fill volume (especially citrate).
-
Separate serum/plasma from cells promptly when required.
B) Urine
Common special-handling scenarios
-
24-hour urine: container, preservatives, accurate timing and documentation
-
Urine culture: sterile container + timely transport (or refrigerate per protocol)
-
Forensic/drug testing: chain-of-custody and sealing
Key handling steps
-
Confirm preservative requirements.
-
Ensure leak-proof containers and clear labels.
-
Apply chain-of-custody rules when results are legally sensitive.
C) Swabs / Respiratory Specimens
Common special-handling scenarios
-
PCR/NAAT: correct swab and transport medium
-
Bacterial culture: site-specific transport media
Key handling steps
-
Standardize swabs/transport kits to reduce errors.
-
Maintain cold chain if testing is delayed.
-
Avoid cross-contamination during collection and processing.
D) CSF / Body Fluids (CSF, pleural fluid, ascites, etc.)
Common special-handling scenarios
-
CSF cell count/culture: extremely time sensitive
-
Special pathogens: higher biosafety precautions
Key handling steps
-
Use STAT delivery pathways.
-
Apply triple packaging and leak protection.
-
Align collection, labeling, and transport steps with biosafety SOPs.
Transport & Packaging: The Most Common Failure Point
As labs become more centralized, transport quality often determines result quality.
Recommended transport standards
-
Clear labeling: specimen type + test + collection time + handling flags (e.g., “Protect from light” / “Keep on ice”).
-
Correct temperature tools: insulated boxes, gel packs, dry ice when required; avoid direct contact that can freeze/hemolyze samples.
-
Triple packaging: primary leak-proof container, secondary sealed container with absorbent, rigid outer packaging.
-
Absorbent material inside the secondary container to manage leaks.
-
Reduced vibration: especially for coagulation/platelet function workflows.
Special Handling Also Depends on Lab Setup (Workflow + Hardware)
Accurate results are not only about SOPs—layout and facilities reduce error rates dramatically.
1) Specimen Receiving Area (Sample Intake)
A well-designed receiving zone improves safety and reduces mislabeling:
-
Dedicated receiving bench for logging, sorting, and scanning
-
Chemical-resistant work surfaces for routine disinfection
-
Clear separation of “clean” vs “potentially contaminated” activities
-
Handwash/sink access and defined waste disposal points
2) Cold Chain Storage Planning
If your test menu includes molecular testing, serology, or outsourced transport:
-
Separate zones for 2–8°C refrigeration, −20°C freezing, and optional −80°C storage
-
Labeled shelving and bin systems to prevent mix-ups
-
Aliquoting space to reduce freeze–thaw cycles and preserve sample integrity
3) Safety & Ventilation
When handling volatile chemicals, fixatives, or strong disinfectants:
-
Ventilation planning (local exhaust / fume hood zones) supports safer workflows
-
Clear chemical-handling areas reduce staff exposure risk and cross-contamination
Tip: If you’re building a diagnostic lab, align your furniture plan with the specimen flow: receiving → prep/aliquot → storage → testing → waste.
CTA:We can help you plan the bench layout, wet area, storage, and fume hood zones based on your test menu and sample workflow.
A Simple “Special Handling” Checklist (Print This)
When a test is ordered, confirm:
-
Correct specimen type
-
Correct tube/container and fill volume
-
Temperature requirement (RT / chilled / frozen / warm)
-
Light protection needed?
-
Max time to lab and time to centrifuge/separation
-
Air exclusion/anaerobic transport required?
-
Biosafety level and packaging required?
-
Chain-of-custody required?
FAQ
1) What’s the most common reason a lab sample is rejected?
Wrong tube type, improper labeling, insufficient volume, and delayed transport are the most frequent causes—especially for coagulation and microbiology specimens.
2) Which tests are most sensitive to transport delays?
Blood gases, lactate, ammonia, coagulation tests (protocol-dependent), CSF cell counts/culture, and platelet function assays are among the most time-sensitive.
3) Why do some samples need protection from light?
Light can chemically degrade certain analytes (such as bilirubin), producing falsely low results even when the patient status is unchanged.
4) Do all PCR/molecular tests require freezing?
No. Many can be refrigerated short-term; freezing is often used for longer storage or delayed shipment. Always follow your kit/lab protocol.
5) How can a lab reduce special-handling errors quickly?
Standardize SOPs, publish a specimen guide at collection sites, use tube-color charts, add handling flags in your LIS, and review rejection trends monthly.
6) What tests require “keep warm” handling?
Some specialized specimens require controlled warm handling to preserve cellular function or prevent precipitation. This is assay-specific—follow the exact lab instruction.
7) What is the difference between refrigerated transport and “on ice” transport?
“On ice” generally means faster cooling and a colder environment than standard refrigeration. Some analytes degrade too quickly for 2–8°C alone.
8) How do I know whether a test needs light protection?
Your specimen guide usually flags photolabile analytes (e.g., bilirubin, porphyrins). When uncertain, protect from light—it’s a low-effort safeguard.
9) Can poor handling cause false positives?
Yes. Contamination in microbiology and molecular workflows can produce false positives, especially when clean/dirty zones and procedures are not separated.
10) What should a small diagnostic lab prioritize first?
Specimen receiving workflow, correct temperature storage, a wet bench with sink, clear labeling, and a safe ventilation plan for chemicals—these reduce errors immediately.
Need help designing a specimen-friendly laboratory?
Share your test menu (biochemistry, hematology, microbiology, PCR, etc.) and room size. We can recommend a practical layout including lab benches, sink stations, storage cabinets, and ventilation zones to support accurate testing and safe handling.

Why We Choose to Be Your Lab Furniture Solution Provider, Not Just a Manufacturer

Max Lab Introduction

Electronics Lab Bench vs Chemistry Lab Bench: What You Need to Know

What Is an Air Shower for Cleanroom?
For After-Sales Support
What do I do if I am not satisfied with my purchase?
We strive to ensure customer satisfaction. If you are not happy with your purchase, please contact us, and we will do our best to resolve any concerns.
For Products
Do you have certifications for your products?
Yes, We have CE, ISO9001, ISO14001, OHSASI18001 and so on.
FAQS
How can I request a quote?
Simply contact us or fill out our online form with your project details. Our team will respond promptly with a free quote.
For Logistics
Do you offer express shipping options?
Yes, we offer express shipping for urgent orders. Please contact our sales team to inquire about availability and additional costs.
For Company
Do you offer our design service from scratch?
Yes, we provide full design support tailored to your lab’s specific needs, including layout planning and furniture customization.

Get in touch with us
If you have any comments or good suggestions, please leave us a message; later our professional staff will contact you as soon as possible.





 Scan QR Code
Scan QR Code
MaxLabFurniture
MaxLab Furniture
daihongada
Max Laboratory